Raman Spectroscopy for knowledgeable people who don’t yet know how awesome Raman spectroscopy is.
Welcome to our page! If you are here, we can assume that you would like to understand what Raman spectroscopy is and how it can be used in your industry or research. Do not worry, we will explain the most important aspects of the technique and how it may help you achieve your goals.
We will build your knowledge step by step, starting from simple concepts. By the end of the series, we will go deeper and provide more precise, technical information.
So, let’s start with the very basics:
What is Raman spectroscopy?
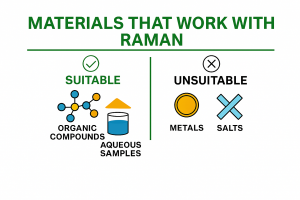
Raman spectroscopy is an analytical technique used to identify materials and study their molecular structure. When you perform a Raman measurement, the instrument records a spectrum that shows the “chemical fingerprint” of the sample.
This fingerprint is unique because every material has its own set of molecular vibrations, and these vibrations produce a specific pattern of peaks in the Raman spectrum.
Raman spectroscopy is used for many applications, with chemical identification and structural analysis being the most common ones. Now that we’ve covered the idea, let’s go a bit deeper into how it works.
How does Raman spectroscopy work?
Many online articles explain the physics in detail, but our goal here is to make it understandable regardless of your academic background. A more advanced version of these chapters will cover the deeper science later.
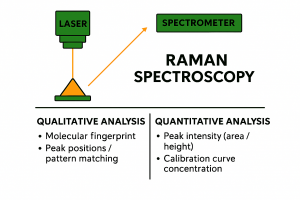
But basically, we have a laser that irradiates a material, this creates a “response” that contains the fingerprint of that material that can be read and analysed by a powerful camera that is attached to a spectrometer.
Let’s begin with the basics.
Raman uses light to identify materials, but not just any light. Raman instruments require monochromatic light, meaning it must have a single, very precise wavelength. This is achieved using lasers with extremely narrow bandwidths (the narrower the better, though also more expensive).
Lasers used in Raman can be different colours (or wavelengths): for example, green (532 nm), yellow (578 nm), or red (630 nm/785 nm).
The interaction with the sample
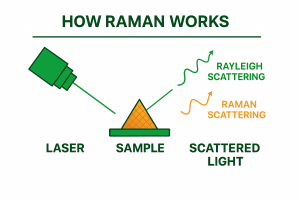
A laser beam is directed onto the sample. When the light hits the molecules:
Most of the light is elastically scattered, this is called Rayleigh scattering. It contains no Raman information.
A very small fraction is inelastically scattered, this is Raman scattering and contains information of the fingerprint of the material we are illuminating with the laser. This is the part we want and we separate to analyse.
During Raman scattering, the energy of the incoming photons interacts with the molecule’s vibrational and rotational states. This interaction causes the scattered photons to shift in energy (sometimes higher, sometimes lower). That shift is what creates the Raman signal.
Collecting the Raman spectrum
The Raman spectrometer rejects the Rayleigh scattering (no information) and collects the scattered light and separates it into its wavelengths, much like a prism splitting white light into colours. A sensitive detector then measures these tiny changes.
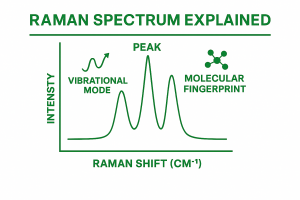
These appear as bands or peaks in a graph, that is what we call the Raman spectrum. Each molecule has its own unique set of vibrational modes, so its spectrum acts like a fingerprint. This is why Raman is often called a “spectroscopic fingerprint” technique.